
Throughout the long history of brewing, beginning in ancient Egypt, Mesopotamia and China, beer was brewed to be consumed quickly after fermentation was complete. The Ancient Egyptians would consume their beer from the fermentation vessel using long reeds as straws.
From a more recent beer history perspective American breweries knew their products would be best enjoyed fresh and various advertising campaigns communicated this fact to their customers.

One of the most popular craft beer styles are IPAs and due to the aggressive hopping of these beers they are best enjoyed very fresh. Stone Brewing fully recognizes this with their Enjoy By IPA beer, displaying the expiration date prominently on the label.

The date shown on the bottle/can of Enjoy By is 35 days after the beer was packaged.
For business reasons most commercially brewed IPAs have advertised best by durations longer than the 35 days reserved for Enjoy By. For example, Stone Brewing uses a suggested best by duration for Stone IPA of 90 days after packaging.
In the 1800’s, the beers brewed in Great Britain which were later be called IPAs were intended to be consumed much later after packaging. But it is important to remember those beers were very different from contemporary IPAs. Those beers of old were principally hoppy from a bittering perspective and were not intended to have prominent hop aroma aspects. In contrast today’s IPA is all about the hop aroma and flavor. And for the current popular substyle of New England IPA also the juicy/hazy quality as well.
There are some beer consumers who like to cellar (age) certain beer styles. A prime example would be a Barley Wine. A Barley Wine is very high in alcohol (e.g., > 8 %ABV) which aids in preserving the beer during the cellaring process. As the beer ages chemical compounds are developed due to oxidation processes which some beer drinkers find to be beneficial and pleasing. A classic flavor which can develop is a flavor reminiscent of sherry. Whether this is truly a beneficial development is subjective to the drinker. Other beer styles which some beer consumers like to age are Belgian Trappist/Abbey Ales, Lambics, Imperial Stouts,…
The majority of beer styles are best consumed fresh with flavors developed from aging considered to be off-flavors.
What happens when a beer ages (i.e., goes stale)?
The chemistry of beer aging is quite involved. Some aspects will be discussed in this article at a cursory level but references will be provided for more in-depth discussion. At the 50,000 foot view what occurs as beer ages is a combination of off-flavor development and reduction of desirable flavors both of which are principally due to oxidation processes.
The classic off-flavor that is often discussed for when a beer gets old is that it develops a flavor described as cardboard or papery. This flavor is from an aldehyde compound called (E) -2-Nonenal which is a result of oxidation. It is generally considered to be more prevalent in old Pale Lagers (e.g., Budweiser, Heineken, Stella Artois). I must confess that I have never perceived a flavor that I would discuss as being cardboard but maybe this is because I do not often drink beers like a Budweiser and when I do I suppose those beers were not too old.
Other examples of beer staling aldehydes and associated off-flavors:
2-Methylpropanal: cheesy
3-Methylbutanal: sweet/fruity
Hexanal: green apple
Furfural: almonds
Methional: sweet, honey-like
Benzaldehyde: bitter almond
Phenylacetaldehyde: cooked potato-like
It is important to keep in mind that how beer staling compounds are perceived is a complex combination of whether the individual compounds are at levels above the taste threshold and how many other compounds exist in total. What is more easily discerned is that a given beer brand will taste different when old vs. when it is fresh.
Also, the compounds discussed above are illustrative for what occurs during the aging of beer and is in no way a comprehensive list. For those interested in additional details I recommend you read the papers referenced at the end of this article.
A classic example of aroma/flavor diminishment is hop fade. As a hoppy beer (e.g., IPA) ages the hop aroma will diminish and with additional aging the flavor will further diminish as well. An IPA that is several months past the packaging date will taste old and tired as compared to a fresh version. Also, with further aging hop bitterness will decrease as well.
Another example of desirable flavor diminishing is that certain ester compounds will decrease with aging. One example is isoamyl acetate which provides the desired banana flavor in the Hefeweizen beer style. As the Hefeweizen ages the flavor of banana will diminish.
Another potential issue with beer over time is that if there are unwanted microorganisms (e.g., bacteria) in the beer the beer can spoil. Large scale breweries (e.g., Anheuser-Busch, etc.) address this potential issue by pasteurizing their packaged beers (i.e., bottles, cans). The packaged beers are passed through a tunnel system where hot water is sprayed on them to heat them up to something like 140 °F (60 °C) for x minutes. The beer industry uses a metric of pasteurization unit (PU) and every minute where the beer is held at 140 °F is defined as 1 PU. So, if the beer was held at 140 °F for 15 minutes that would be 15 PUs of treatment. As will be further discussed below exposing beer to higher temperatures accelerates aging/staling so the decision to pasteurize beer is a tradeoff between reducing the potential for spoilage and accelerating aging/staling. Implementing pasteurization is costly and consequently most craft breweries do not pasteurize their beers, they depend upon good sanitation processes within the brewery to mitigate unwanted microorganisms within their beers.
All of the changes discussed above is due to oxidation which occur as a combination of the oxidative chemical reactions that occur over time. At a high level the ‘solution’ is easy: reduce oxygen and reduce time. But wait there is more:
Svante August Arrhenius – a very smart Chemist

I took chemistry in high school and three terms in college but I have zero recollection of being taught about Arrhenius and his famous (well, not to me back in the day) equation. The Arrhenius Equation is not an equation like E=mc2 but more like a word association thing: for every 10 °C (18 °F) increase in temperature, a chemical reaction will proceed two to three times more quickly. Or looking at it from a cold storage perspective: for every 10 °C (18 °F) decrease in temperature, a chemical reaction will proceed two to three times more slowly. For a beer scientist this is a “Eureka!” moment. If you want to slow down beer aging (i.e., beer going stale) colder temperatures will slow down those nasty oxidation processes.
Below is a handy graph courtesy of Dr. Charlie Bamforth which illustrates how cold storage improves the shelf like of beer. Keep in mind the temperature is in Celsius.
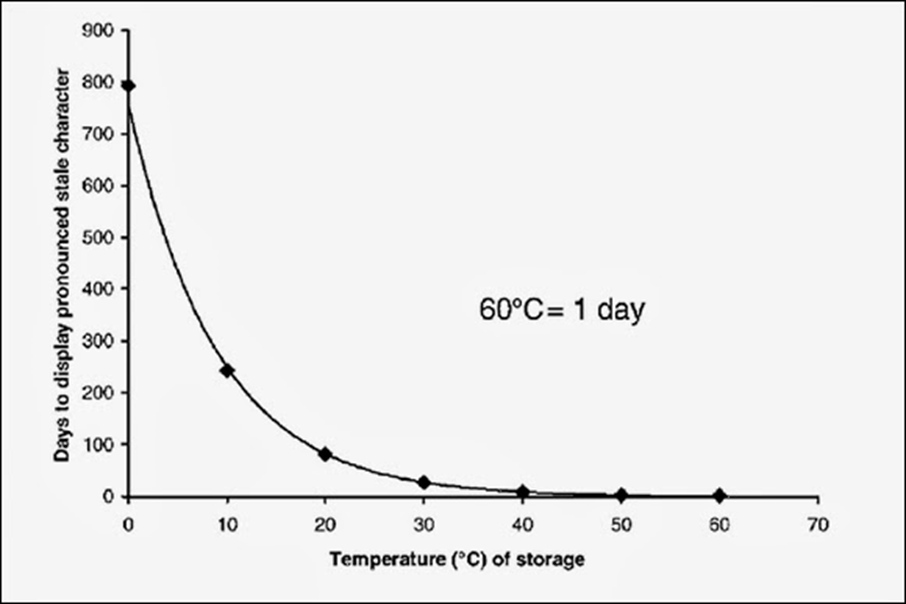
Since minimizing oxygen in the beer will reduce oxidation processes there is a clear goal to brewers: keep oxygen out as much as possible.
A metric that is used in the commercial brewing industry is Total Packaged Oxygen (TPO). TPO is a combination of the dissolved oxygen (DO) within the beer that enters the package (bottle, can) and the amount of oxygen in the headspace of the package. The TPO can be measured using an oxygen level meter. One analytical instrument does this by measuring both the headspace oxygen and the DO in the beer and computes the TPO. A good industry target for TPO is 50 ppb. Needless to say, not all commercial breweries achieve here.
For homebrewers who keg their beers an optimum practice is to conduct a closed transfer of the beer to the serving keg. A good discussion on how to conduct closed transfers, including a video, is available here:
https://www.brew-dudes.com/how-to-closed-transfer-homebrew/7576
One aspect to keep in mind is that purchased tanks of CO2 used for force carbonation are not 100% pure; a typical tank will be 99.9% CO2. The remaining 0.1% will be a combination of other stuff including oxygen. An alternative strategy to carbonate beer in a keg is via a spunding valve which results in natural carbonation thus eliminating the extra oxygen that comes from purchased tanks of CO2.
For homebrewers who bottle their beers the good news is that bottle conditioning decreases oxygen levels during the two week conditioning phase since oxygen is consumed during the secondary fermentation. Also, during the secondary fermentation sulfur dioxide (SO2) is developed and this compound is a potent antioxidant. After the beers are carbonated the presence of live yeast within the bottles operate like antioxidants in that they will continue to process oxygen which will ingress through the bottle cap liners over time. This aspect was quantified by Edward E. Morris as part of his Master’s Thesis entitled “Antioxidant Potential of Yeast Containing Beer”. Edwards analyzed bottles of artificially carbonated beer, bottle-conditioned beers and artificially carbonated beers with live yeast added over time (up to 6 months).
From the thesis: “Antioxidant potentials were determined using Ferric Reducing Antioxidant Potential (FRAP) analysis.” Three different yeast strains were studied: California Ale Yeast (WLP001), San Francisco Lager Yeast (WLP810), and Belgian Wit Ale Yeast (WLP400). A result of the study is that yeast strain type is a variable (results varied with each strain).
One summary result from the thesis: “The bottle-conditioned beers had the greatest overall antioxidant activity”.
One other strategy which could be employed is the addition of antioxidant chemical compounds. Some examples are sodium metabisulfite, potassium metabisulfite, and ascorbic acid (vitamin C). I personally have no experience with these compounds. Based upon my readings these compounds should be used judiciously; for example, if too much potassium metabisulfite is used it can cause a sulphury taste (e.g., rotten eggs) in the beer.
Another Chemist which I never learned about (or I forgot?) is Henry John Horstman Fenton (whew, what a long name!). Mr. Fenton is the person who discovered a chemical reaction which now bears his name: Fenton reaction. A short description via the Science Direct website:
“The Fenton reaction was described for the first time by Fenton, who observed the oxidation of tartaric acid by hydrogen peroxide in the presence of ferrous ions (Fenton, 1894).”
The important words above are “ferrous ions” (otherwise known as iron ions) and “oxidation”. When it comes to beer the issue is that various metal ions (e.g., iron, copper and manganese) present in the wort/beer can promote oxidation/staling.
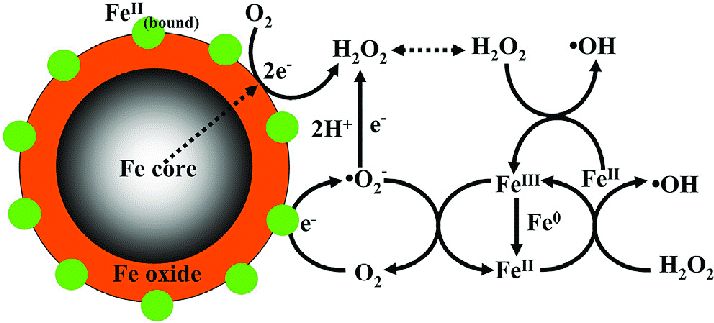
These metal ions can be removed by complexation through the use of chelating agents/chelators. One class of chelating agent are tannic acids. While attending HomebrewCon 2019 I attended a presentation by Joe Formanek where he discussed a tannic acid product called Brewtan B. Below is how Brewtan B is detailed by the manufacturer Ajinomoto:
“Brewtan B is a 100% natural gallotannin specially designed for the brewing industry. This tannin improves the clarity and shelf life of beer by reacting with haze-sensitive proteins (proteins rich in proline) and SH-group-containing proteins by adsorption and precipitation. They also chelate iron, thereby inhibiting the Fenton oxidation reaction.
Brewtan B:
Used in the mash tun as iron chelator (2–6 ppm)
Used in brew kettle for kettle fining (2–6 ppm)”
One aspect worth highlighting is that Brewtan-B also operates like a clarifier.
Since attending Joe Formanek’s presentation I have incorporated using Brewtan B in my homebrewing for non-hoppy beer styles. I add some Brewtan B to the water for the mash and also in the kettle for the last 15 minutes of boil (with rehydrated Irish Moss flakes one minute afterwards). In discussion with Joe at the presentation he made mention that he discourages the use of Brewtan B for hoppy beer styles (e.g., IPAs) since this product tends to ‘muddle’ the hop aroma/flavor.
One other aspect that leads to accelerated beer aging (hop fade) is rough handling of the beer in transport.
Below is an interesting discussion about how shipping can impact beer via Tom Nielsen of Sierra Nevada Brewing:
“According to Nielsen, agitation during shipping can be a significant contributing factor to degradation in aroma. As a beer sits on delivery trucks and eventually finds its way to your local liquor store, the beer’s aromas can be kicked up through the head space and slowly forced out of the crown liner, a process Sierra Nevada refers to as scalping. Nielsen also says oxygen will destroy hop aroma very quickly, whether naturally over time, or through the bottling process.
“We’ve found the hop aroma of a fresh beer shipped overnight from Boston compared to the same beer that just sat here in Chico was very much reduced,” said Nielsen. “This degradation doesn’t noticeably impact bitterness. But since aroma plays a significant role in your perception of taste, it can greatly influence your overall enjoyment of the beer.”
One of the benefits of homebrewing is that the beers we consume at home do not have to suffer from the negative effects of rough handling. For distributing breweries there is no getting around the fact that beer will suffer from vibration/agitation during transport: trucks, trains, ships, etc.
A more scientific discussion of the effects of vibration/agitation is provided in the below referenced article “Influence of transport and storage conditions on beer quality and flavour stability”.
As discussed previously cold beer has an increased shelf life. So, the ‘simple’ answer here is always store beer cold. This is not always an easy implementation for commercial breweries. I can anecdotally report in my area Lawson’s Finest Liquids (what a cool name for a brewery) is able to ensure that their beers are continuously stored cold at my local (Philadelphia area) beer retailers. My presumption is that this is a requirement in the agreement that Lawson’s has with the local Wholesale Distributor; that once the beer is delivered to the area it must be continuously stored cold and it is the responsibility of the Wholesale Distributor to enforce this aspect with their customers (beer retailers). I can think of no other commercial breweries who do the same.
As homebrewers we have it within our control as to how we want to store our beers. Homebrewers who keg can store those kegs in their refrigerators/keezers. Homebrewers who bottle can place their beers in a dedicated beer refrigerator or just depend upon the anti-oxidative power of the live yeast to mitigate beer staling.
An additional consideration for storage of bottled beers is that they are best stored in a dark place (preferably in your dedicated beer refrigerator). Bottles exposed to light over time may become lightstruck (i.e., skunked). The skunky aroma/favor is a result of the development of a mercaptan compound (3-methyl-2-butene-1-thiol).
With just a few exceptions beer is best enjoyed fresh. As beer consumers there are many steps we can take to ensure we are drinking beer in the peak of condition.
Homebrewing is a great option since we have full control here. Once the homebrewed beer is packaged and given a short span of time if bottle conditioning the beer is fresh and in optimum condition for enjoyment. And since we homebrewers do not distribute our beers to unknown and far flung places we do not have to worry that our beers are getting ‘beat up’ by high heat, getting shaken or other nasty conditions.
When it comes to drinking commercially brewed beers a good strategy is to visit local breweries and drink beer on draft at the source. It is generally speaking a reasonable expectation that the draft beer there has been brewed and packaged a short time ago and have been continuously stored cold. You can even purchase packaged (e.g., canned) beer there to take home but it still would be prudent to check for a packaging date prior to purchase.
When purchasing commercial beer at beer retailers (e.g., supermarkets, convenience stores, liquor stores, etc.) the best strategy is to check dates prior to purchase (with the best date being a packaged-on date). Use your judgement on how long after packaging a given beer brand is ‘good’ for. A good rule of thumb is that hoppy beer like IPAs is best purchased very fresh (I personally refuse to purchase IPAs that are over 3 months old) and other beer styles like Stouts/Porters can typically be ‘good’ for a longer duration (e.g., 6 months). The intangible here is that in the absence of knowing what the quality of a given brewery’s packaging line is (e.g., what is the TPO of a beer brand) you just have a date to make a purchasing decision. Some beer consumers tend to gravitate to the beer refrigerators to purchase those beers (and cold beer is indeed better) but you never really know how long those beers were stored cold at the retailer; maybe that x month old beer was just placed in the refrigerator a day earlier and was warm on the shelf the majority of that timeframe.
Another potential consideration when purchasing commercial beer is package type. Canned beer has the advantages that cans are totally impervious to both oxygen ingress and light. In contrast a bottled beer will permit oxygen to ingress through the plastic cap liner over time. How much oxygen ingresses into a bottle over time are dictated by the laws of physics (i.e., Dalton’s law of partial pressures, Fick's laws of diffusion) and the oxygen permeability coefficient of the specific material used to make the cap liner. For example, if a given beer brand is available in both bottles and cans and those two products had the same TPO value at comparable packaging dates, the canned product would be the preferable choice
Now, go out and brew some fresh homebrew! And if you have one, place those beers in a dedicated beer refrigerator.
The above article is an overview. For people interested in the detailed science below are some technical articles I recommend for further reading:
Bart Vanderhaegen, Hedwig Neven, Hubert Verachtert, and Guy Derdelinckx, “The chemistry of beer aging—a critical review” Food Chemistry 95 (2006), 357–81.
Barbara Jaskula-Goiris, et. al., “Influence of transport and storage conditions on beer quality and flavour stability” Journal of the Institute of Brewing 125 (2019), 60-68.
All contents copyright 2024 by MoreFlavor Inc. All rights reserved. No part of this document or the related files may be reproduced or transmitted in any form, by any means (electronic, photocopying, recording, or otherwise) without the prior written permission of the publisher.